20
15
10
5
0
40
30
20
10
0
10
20
30
Temperature (C)
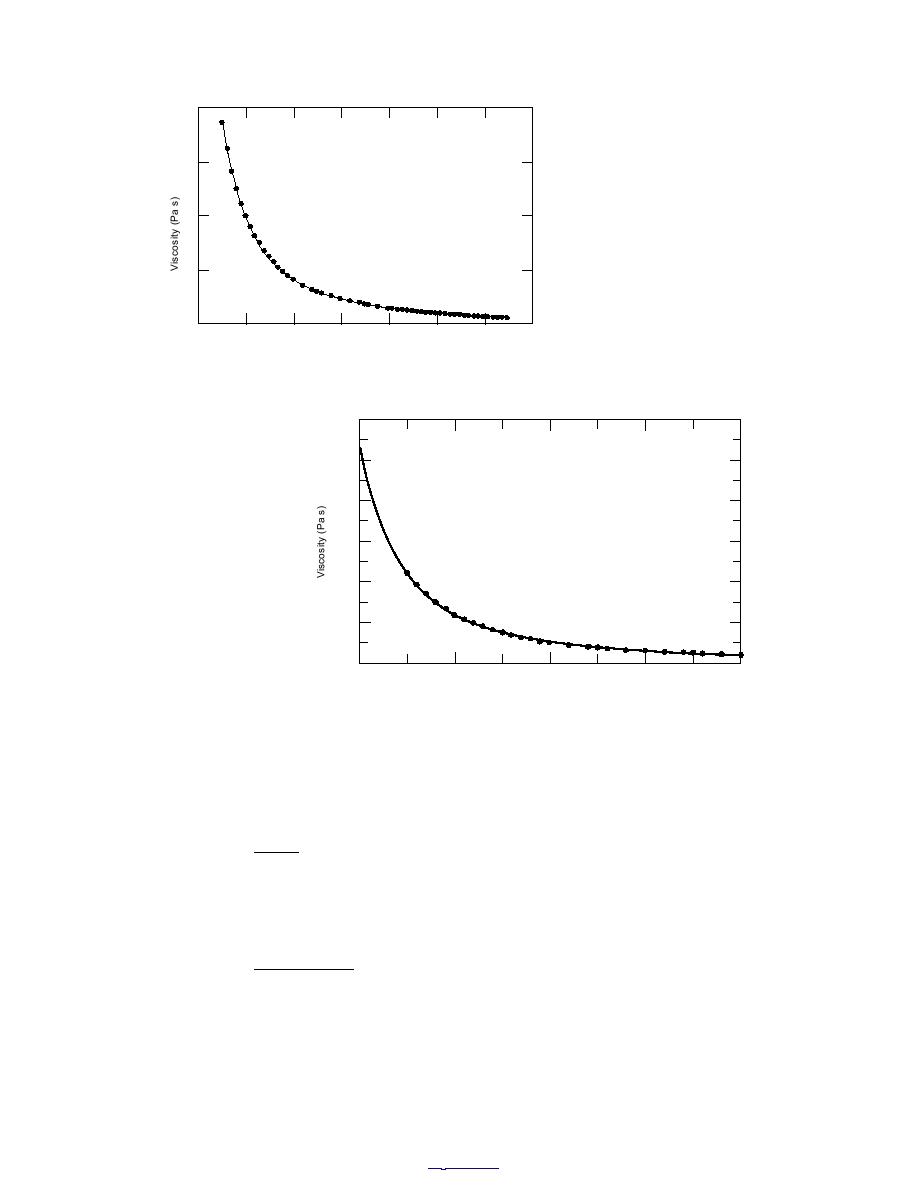
Figure 3. Supercooled water viscosity modeled by modified
VogelTammannFulcher (VTF) equation.
50
40
30
20
10
0
30
20
10
0
40
Temperature (C)
Figure 4. Supercooled water viscosity estimated by Speedy's (1987)
empirical formula.
In addition to temperature, the viscosity of aqueous solutions is affected by the type and
concentration of solutes. These effects are discussed in standard references (e.g., Harned
and Owen 1958, Robinson and Stokes 1959). Vand (1948) derived an equation
A3c
ln vrel =
(7)
1 - Q′c
where νrel is the ratio of the solution's viscosity to that of the water.
This author found that an extension of the Vand equation gave improved fits to viscosi-
ties of NaCl solutions:
( A0 + A1T )c .
ln vrel =
(8)
1 - (Q0 + Q1 T )c
′
′
This equation was fitted to the data of Lyle and Hosking (1902), who reported the viscosity of
NaCl solutions with initial concentrations (at 15C) of 0.1, 0.2, 0.5, 1.0, 2.0, and 4.0 mol dm3.
The following parameter estimates (and standard errors of the estimates) were obtained:
′
′
A0 = 0.116 90 (0.003 97), A1 = 0.000 579 (0.000 012), Q0 = 0.440 930 (0.003 467), and Q1 =
0.001 065 909 6 (0.000 000 000 00).
7
TO CONTENTS




 Previous Page
Previous Page
